














| Still deciding? Get samples of $ ! US$ 0/Piece Request Sample |
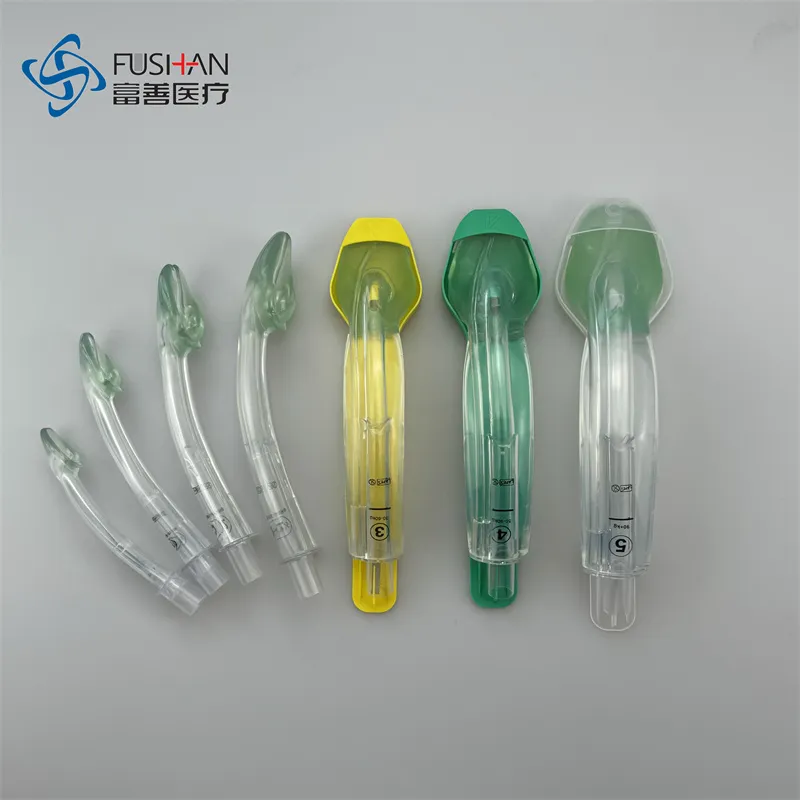
The laryngeal mask is used for general anesthesia surgery to ensure the breathing passage of patients. It effectively seals the laryngeal cavity, preventing secretions from entering and impacting respiratory function under general anesthesia. The central laryngeal mask features a breathing passage connected directly to the laryngeal cavity, with the distal end compatible with ventilators. Unlike endotracheal intubation, this laryngeal mask is inserted through the larynx to avoid injury to the cavity, dislocation of the arytenoid joint, and damage to the tracheal mucosa. It is suitable for most general anesthesia operations except for upper respiratory tract procedures.
ALMA-G is a single-use supraglottic airway management device crafted from high-quality thermoplastic elastomer. It features:
| Size | Patient Weight | Max Size of Gastric Tube |
|---|---|---|
| 1.0 | <5 KGS | N/A |
| 1.5 | 5-12 KGS | 8Fr |
| 2.0 | 10-25 KGS | 8Fr |
| 2.5 | 25-35 KGS | 12Fr |
| 3.0 | 30-60 KGS | 12Fr |
| 4.0 | 50-90 KGS | 12Fr |
| 5.0 | ≥90 KGS | 12Fr |



The factory covers an area of 13,000 sqms. Both class 10,000 and class 100,000 clean workshops are designed and constructed according to GMP standards. Equipped with precise manufacture and inspection facilities, the facility produces various medical devices made of silicone, polyurethane (PU), and polyvinyl chloride (PVC). Professional labs are available for sterile, physicochemical, and electrical tests, alongside a CE verified ETO sterilization chamber.






 Ryu Medical
Ryu Medical