

1 / 5










| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |

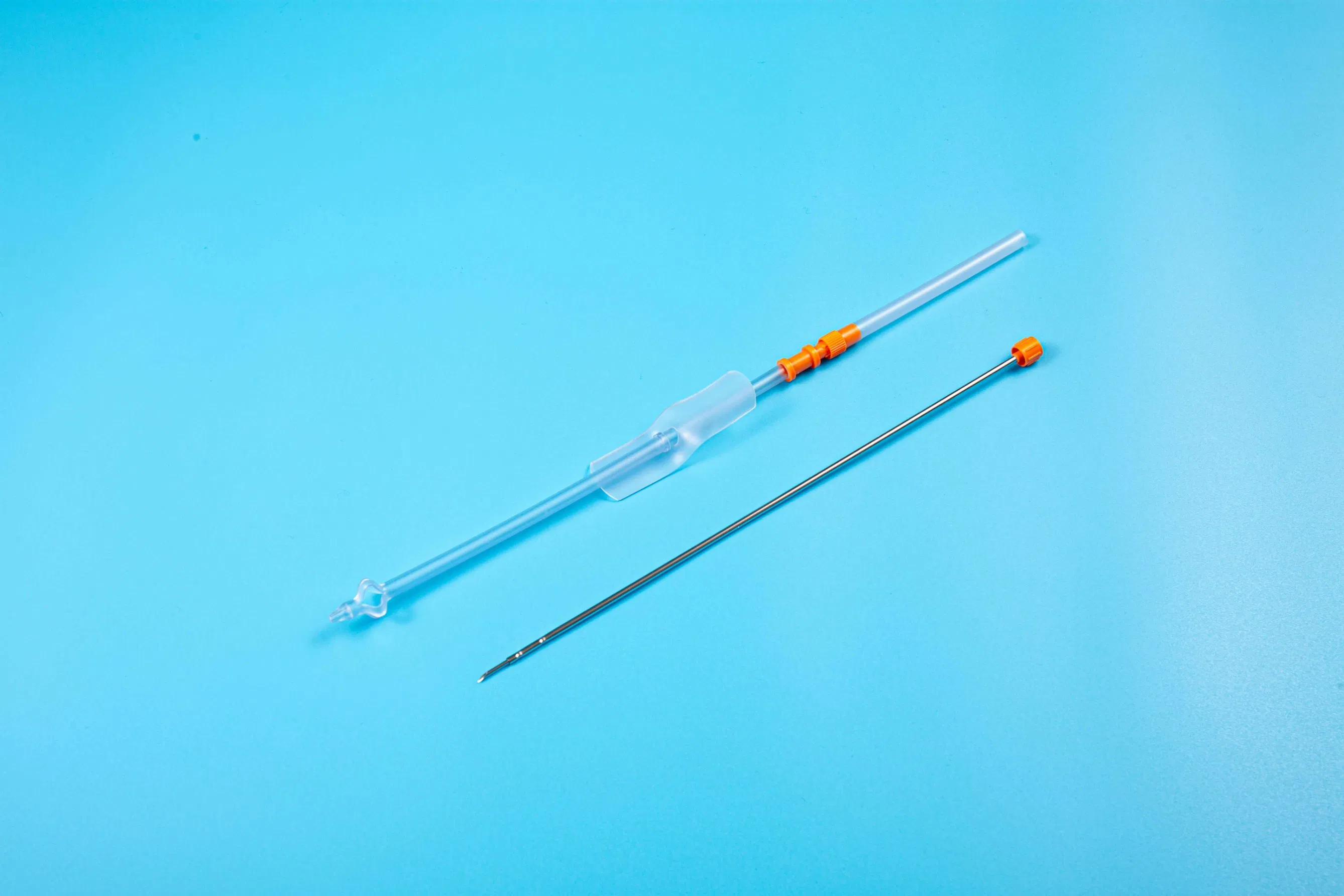
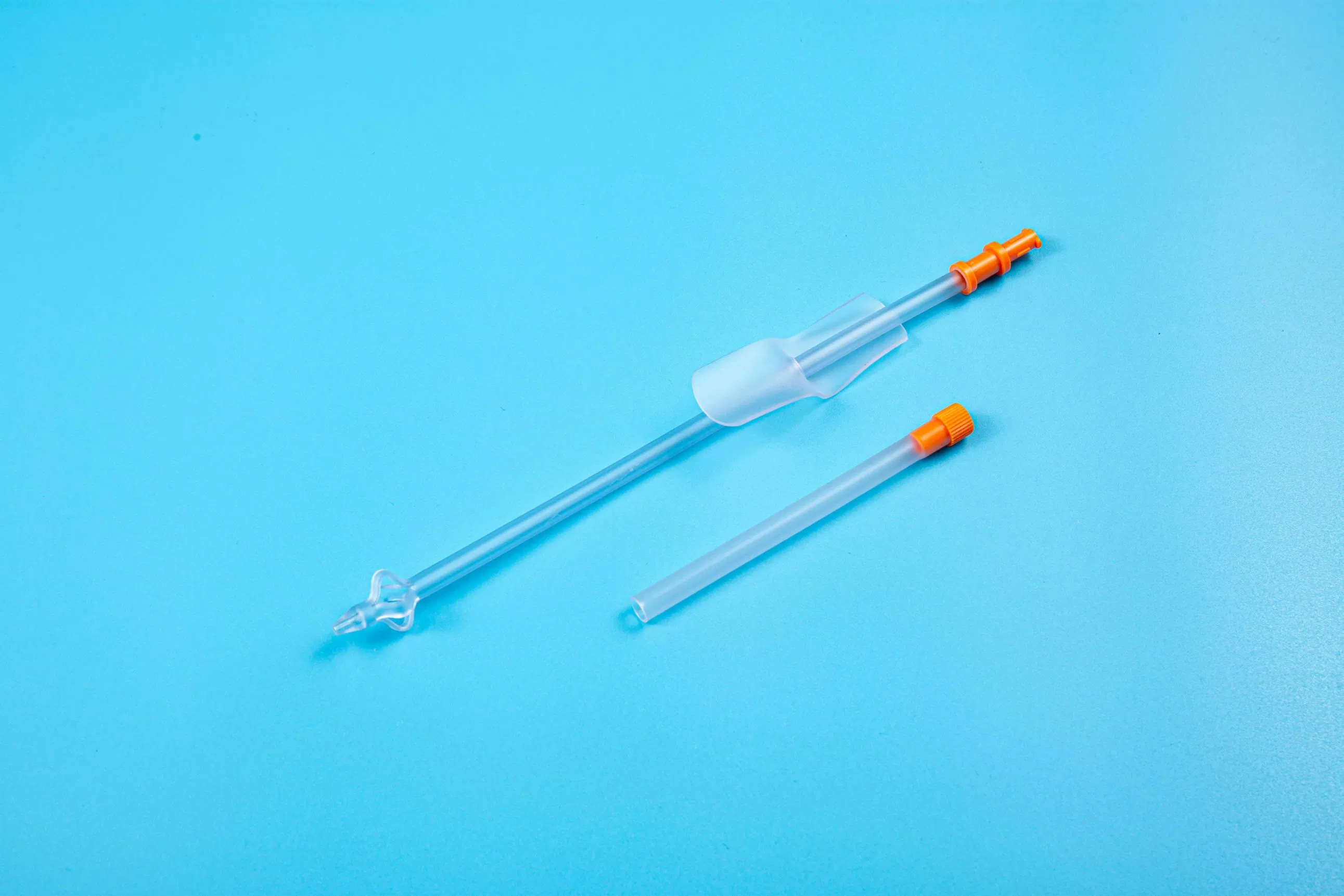
This medical device is applied for temporary urinary diversion, suprapubic bladder puncture fistula, and direct drainage of urine from the bladder.
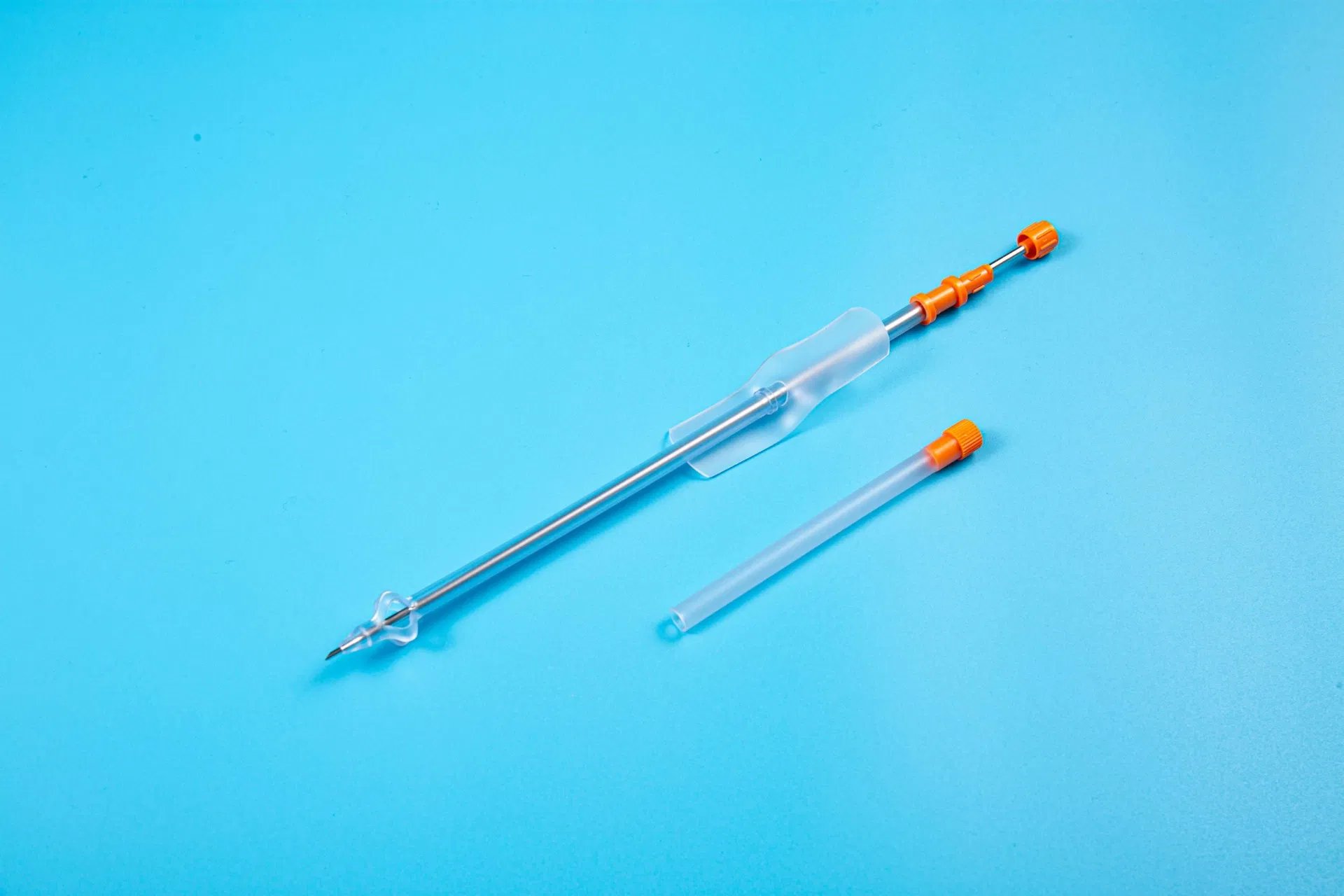
| Product Name | Disposable Sterile Suprapublic Cystostomy Catheter |
| Category | Surgical Supplies Materials |
| Sterilization | Ethylene Oxide (EO) Sterilization |
| Length | 200mm |
| Specifications | F14, F18, F24 |
| Validity | 3 Years |
| Packing | Plastic Sterile Containers |
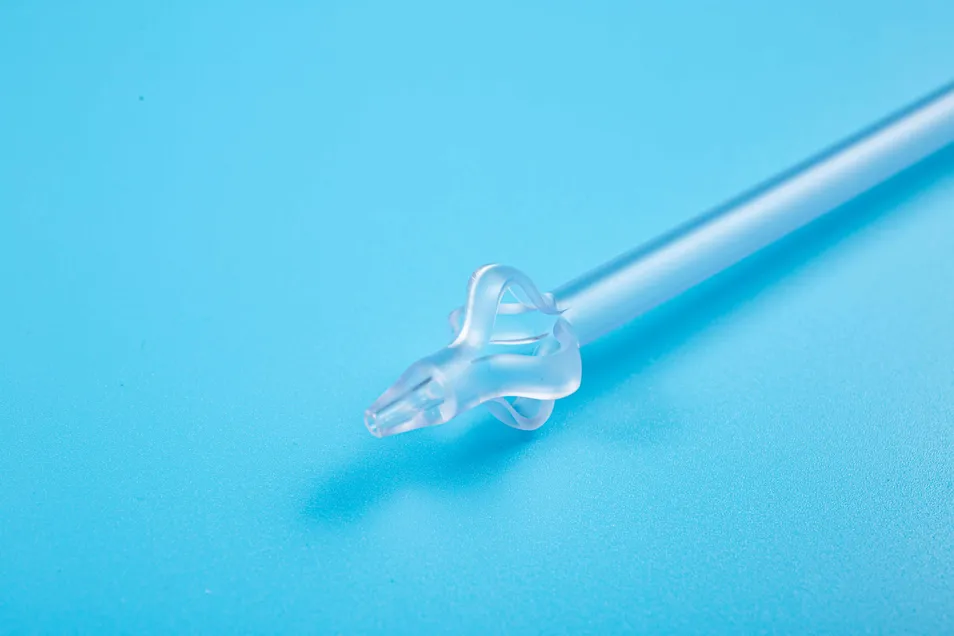
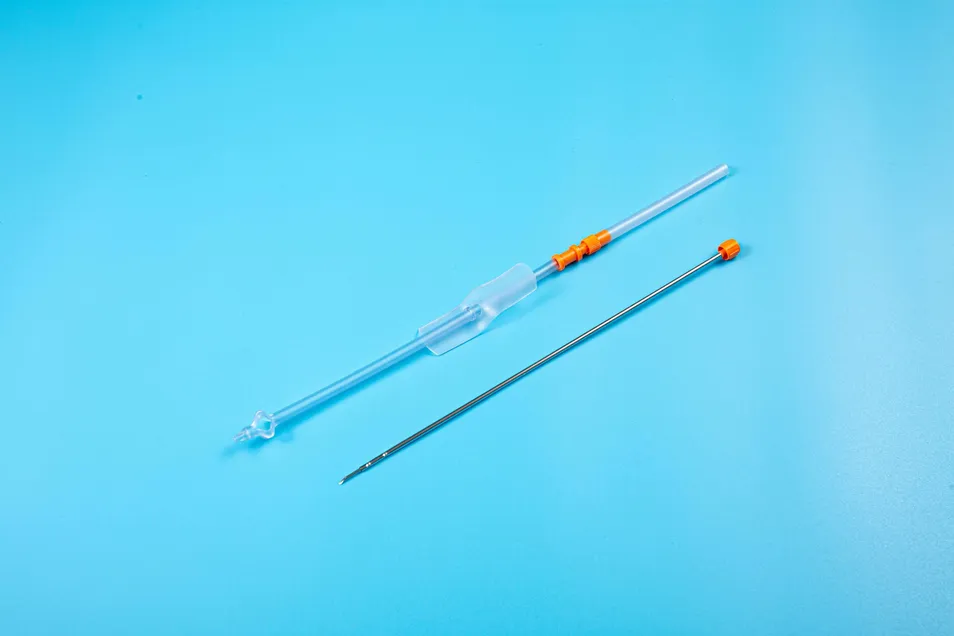

Target Audience: Suitable for patients with acute urinary retention, penile, or urethral injury.
Contraindications: There are no absolute contraindications for this product.



 Ryu Medical
Ryu Medical