



1 / 5










| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |

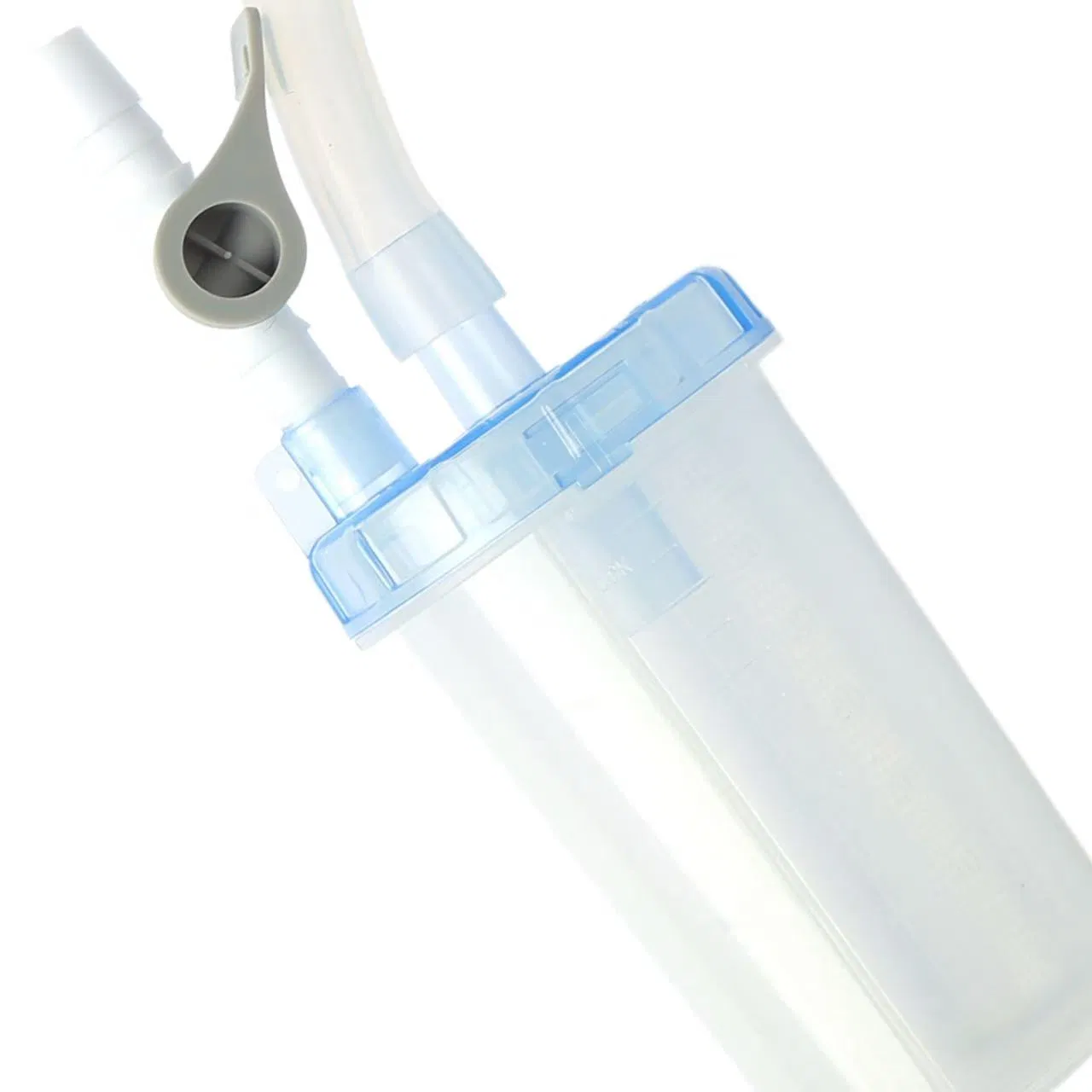
The Negative Pressure Drainage Device is a precision-engineered medical tool composed of a high-quality barrel, filter screen, secure cap, sealing ring, specialized valve, and connecting tube.
Designed to effectively prevent cross-contamination. By integrating the barrel with the drainage system via secure tubing, it enables effortless fluid extraction, significantly reducing clinical procedure time and optimizing surgical workflow efficiency.
The high-transparency housing allows healthcare professionals to visually monitor the characteristics of drained fluids in real-time. This can be performed without opening the system, providing critical data for immediate clinical assessment and intervention decisions.
| Technical Specifications | |
|---|---|
| Product Name | Negative Pressure Drainage Device |
| Sterilization Method | EO (Ethylene Oxide) |
| Available Lengths | 300mm, 350mm, 400mm, 450mm, 500mm, 550mm, 600mm |
| Standard Capacity | 140ml |
| Main Application | Minimally Invasive Surgical Drainage |
| Material Package | Medical Grade Plastic Package |
| Stock Status | Available for Immediate Supply |





 Ryu Medical
Ryu Medical