Basic Information
Certification
CE, ISO13485
Sterilization
Ethylene Oxide (EO)
Application
Hospital / Emergency
Capacity
100,000 PCS/Month
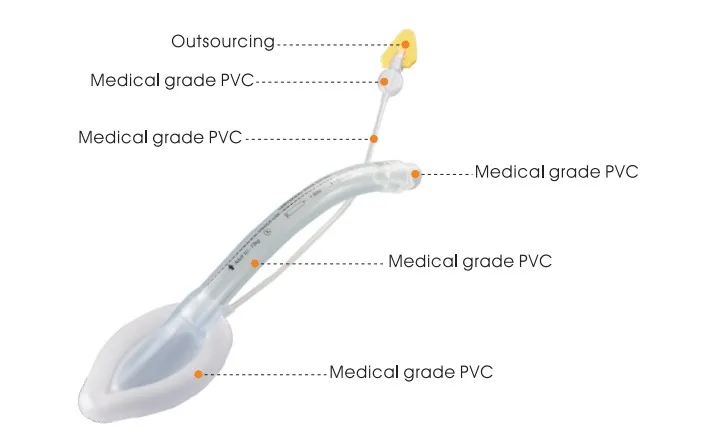
Material
Medical-grade Silicone / PVC
Product Description
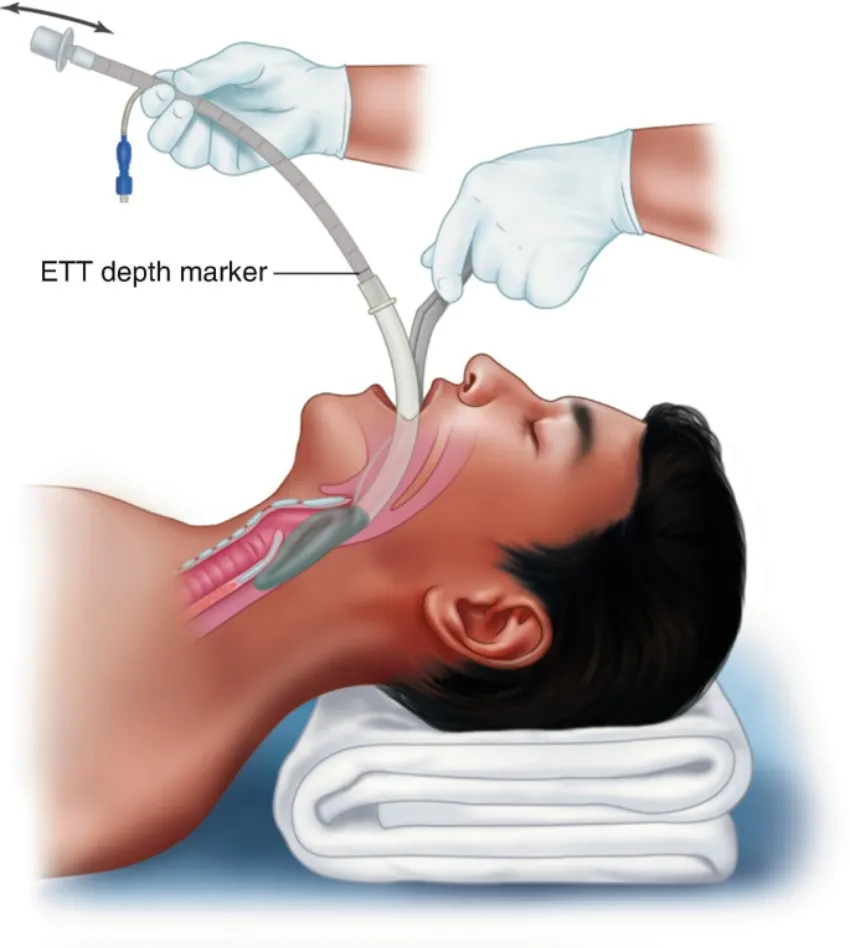
Fiberoptic Ready LMAs for Difficult Airway Management and Guided Intubation
Our Advanced Supraglottic Airway Device (SAD) is designed to provide a secure and efficient airway solution for anesthesia and emergency airway management. It offers superior sealing performance, reduced airway trauma, and easy insertion.
| Item |
Description |
| Product Name | Advanced Supraglottic Airway Device |
| Material | Medical-grade Silicone / PVC |
| Type | Reusable or Disposable |
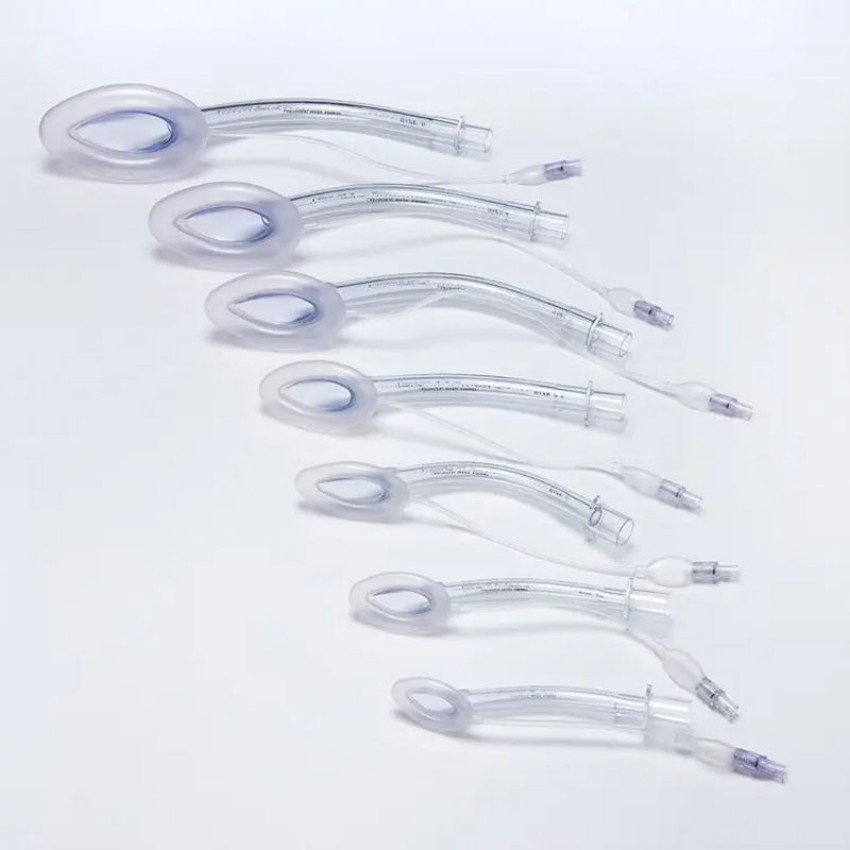
| Size Options | 1#, 1.5#, 2#, 2.5#, 3#, 4#, 5# |
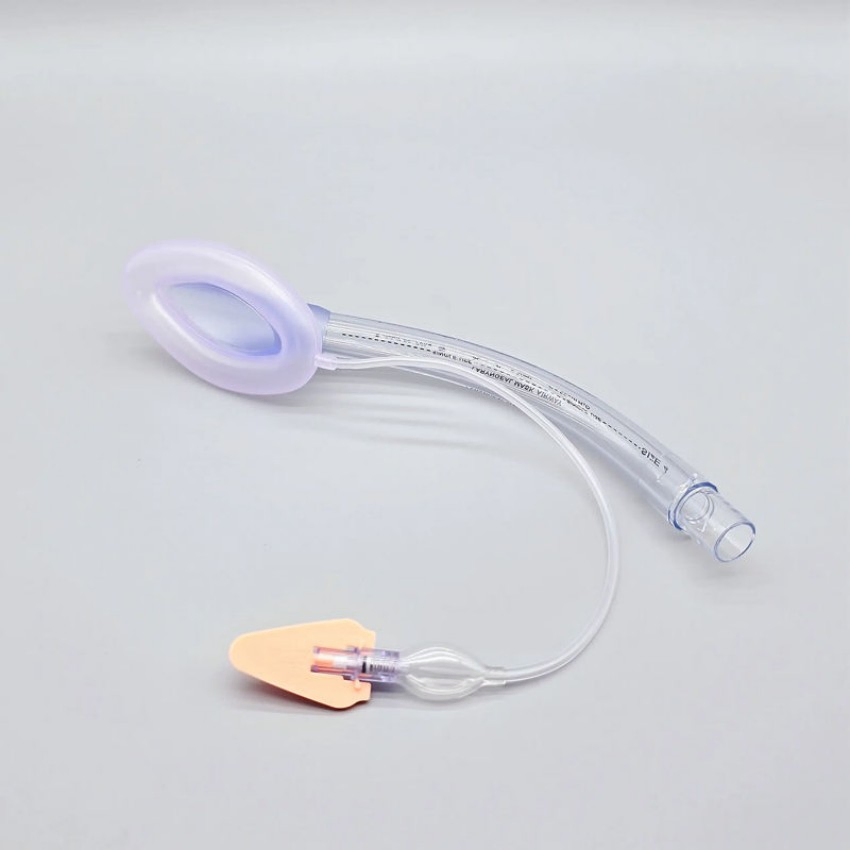
| Connector | Standard 15mm ISO Connector |
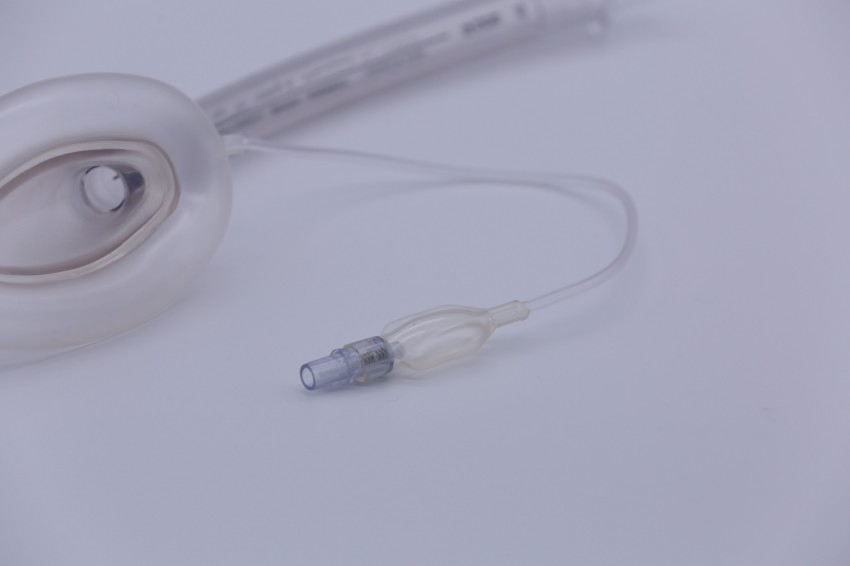
| Cuff Type | Inflatable or Non-inflatable |
| Sterilization | EO Gas Sterilized or Autoclavable |
Key Features
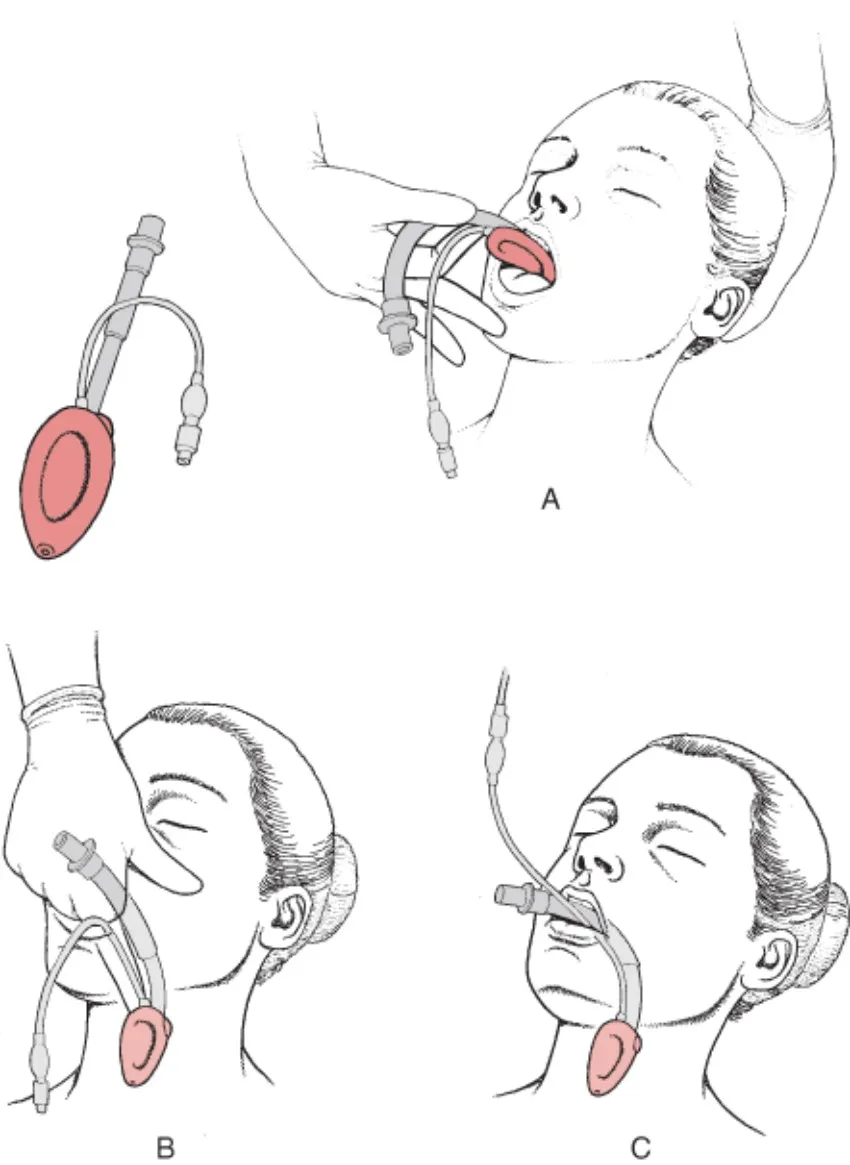
- Anatomically Designed Shape - Optimal fit and minimal resistance.
- Soft, Smooth Cuff - Reduces mucosal injury risk.
- Easy and Rapid Insertion - Critical for emergency efficiency.
- High Seal Pressure - Suitable for controlled ventilation.
- Latex-Free & Biocompatible - Minimizes allergic reactions.
- Universal Sizes - Options from neonates to adults.
Advantages & Applications
Clinical Benefits:
- Enhanced patient comfort and airway protection.
- Simplified training for medical staff.
- Cost-effective alternative to standard intubation.
Usage Scenarios:
- Operating rooms for general anesthesia.
- Emergency departments and ICU.
- Ambulatory surgical centers.
Precautions & Packaging
- Use only by trained medical professionals.
- Check device integrity before use; do not use if packaging is damaged.
- Single-use models must not be reused.
- Avoid over-inflation of the cuff to prevent mucosal injury.
- Packaging: Individually packed in sterile blister bags (50-100 pcs/carton).
Frequently Asked Questions
What materials are the Supraglottic Airway Devices made of?
They are manufactured from high-quality, medical-grade silicone or PVC, ensuring they are biocompatible and latex-free.
Are these devices reusable or disposable?
We offer both options. Disposable models are EO gas sterilized for single use, while reusable models are designed to be autoclavable.
How do I choose the correct size for a patient?
Sizes range from 1.0# to 5.0#, covering neonates, infants, children, and adults. Selection is typically based on the patient's weight as per clinical guidelines.
Can these be used for difficult airway management?
Yes, they are Fiberoptic Ready and specifically designed for difficult airway management and guided intubation scenarios.
What is the typical shelf life of the product?
The shelf life is generally 3 to 5 years, provided they are stored in a cool, dry environment away from direct sunlight.
Do the devices comply with international standards?
Yes, our devices are CE and ISO13485 certified, and the connectors follow standard 15mm ISO requirements.













 Ryu Medical
Ryu Medical