



1 / 5










| Still deciding? Get samples of $ ! US$ 10/Piece Request Sample |

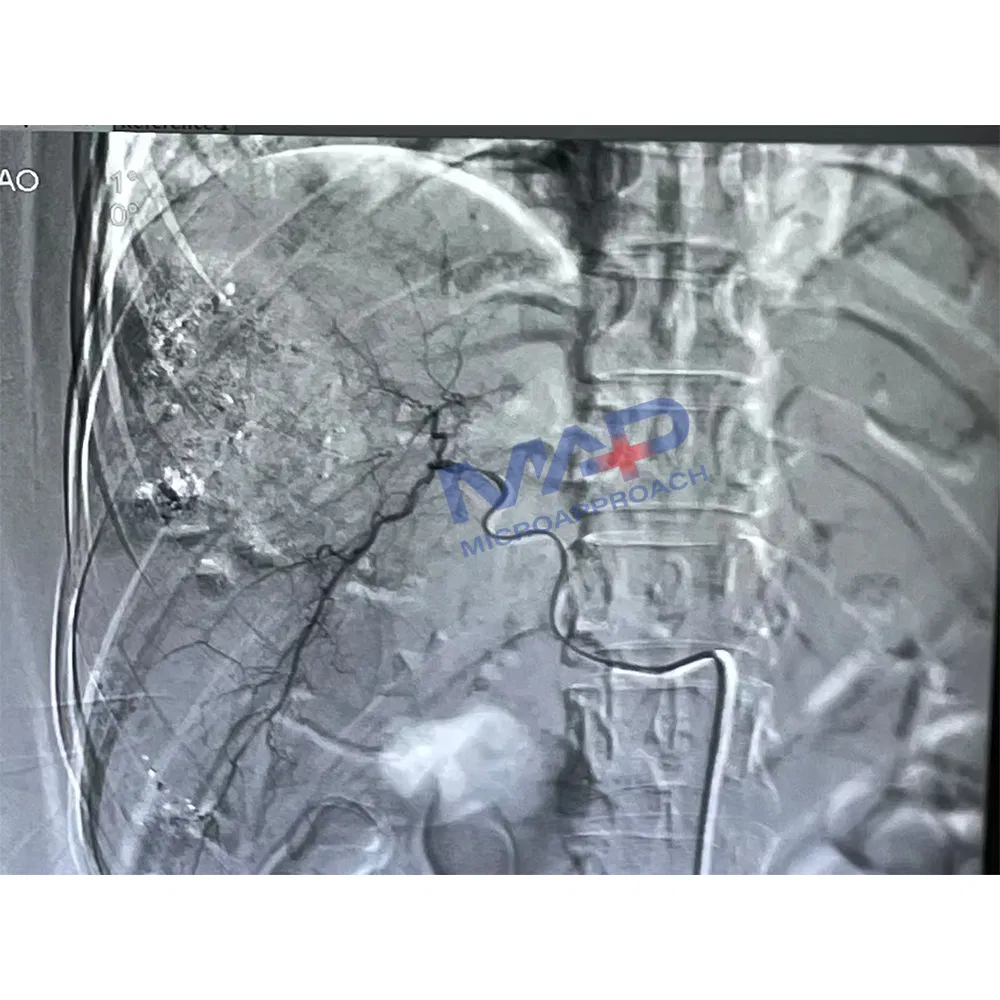
The Micro Catheter features a flexible distal region with a hydrophilic coating applied to the distal outer surface to ensure smooth navigation. A radiopaque marker is positioned proximal to the tip to facilitate clear fluoroscopic visualization during procedures. The proximal end incorporates a standard luer adapter for easy attachment of accessories, and a tip straightener is included.
This device is specifically designed for injecting diagnostic agents (contrast agents) and therapeutic agents (pharmaceuticals, embolic materials), while supporting guidewires within the peripheral or coronary vasculature.

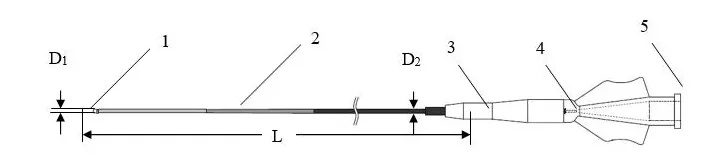
Fig 2-1: Basic structure diagram of Micro Catheter
| Model | Distal OD (Fr/mm) | Proximal OD (Fr/mm) | Length (cm) | Guide Wire (in/mm) | Marker |
|---|---|---|---|---|---|
| SMC-1.8-130 | 1.8/0.6 | 2.7/0.9 | 130 | 0.018/0.46 | 1 |
| SMC-1.8-150 | 1.8/0.6 | 2.7/0.9 | 150 | 0.018/0.46 | 1 |
| SMC-2.0-130S/D/T | 2.0/0.67 | 2.7/0.9 | 130 | 0.018/0.46 | 1/2/3 |
| SMC-2.0-150S/D/T | 2.0/0.67 | 2.7/0.9 | 150 | 0.018/0.46 | 1/2/3 |
| SMC-2.8-130S/D/T | 2.8/0.93 | 2.8/0.93 | 130 | 0.021/0.53 | 1/2/3 |
| SMC-2.8-150S/D/T | 2.8/0.93 | 2.8/0.93 | 150 | 0.021/0.53 | 1/2/3 |



 Ryu Medical
Ryu Medical