













| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |
| Model NO. | Negative Pressure Drainage Device |
| Sterilization | Ethylene Oxide (EO) Sterilization |
| Length | 300mm - 600mm |
| Specifications | 12×300, 12×350, 12×400, 12×450, 12×500, 12×550, 12×600 |
| Capacity | 140ml |
| Certification | ISO13485 |
| Classification | Class 1 / Class II |
| Validity | 3 Years |
| Usage | Minimally Invasive Surgical Materials |
| HS Code | 9018390000 |




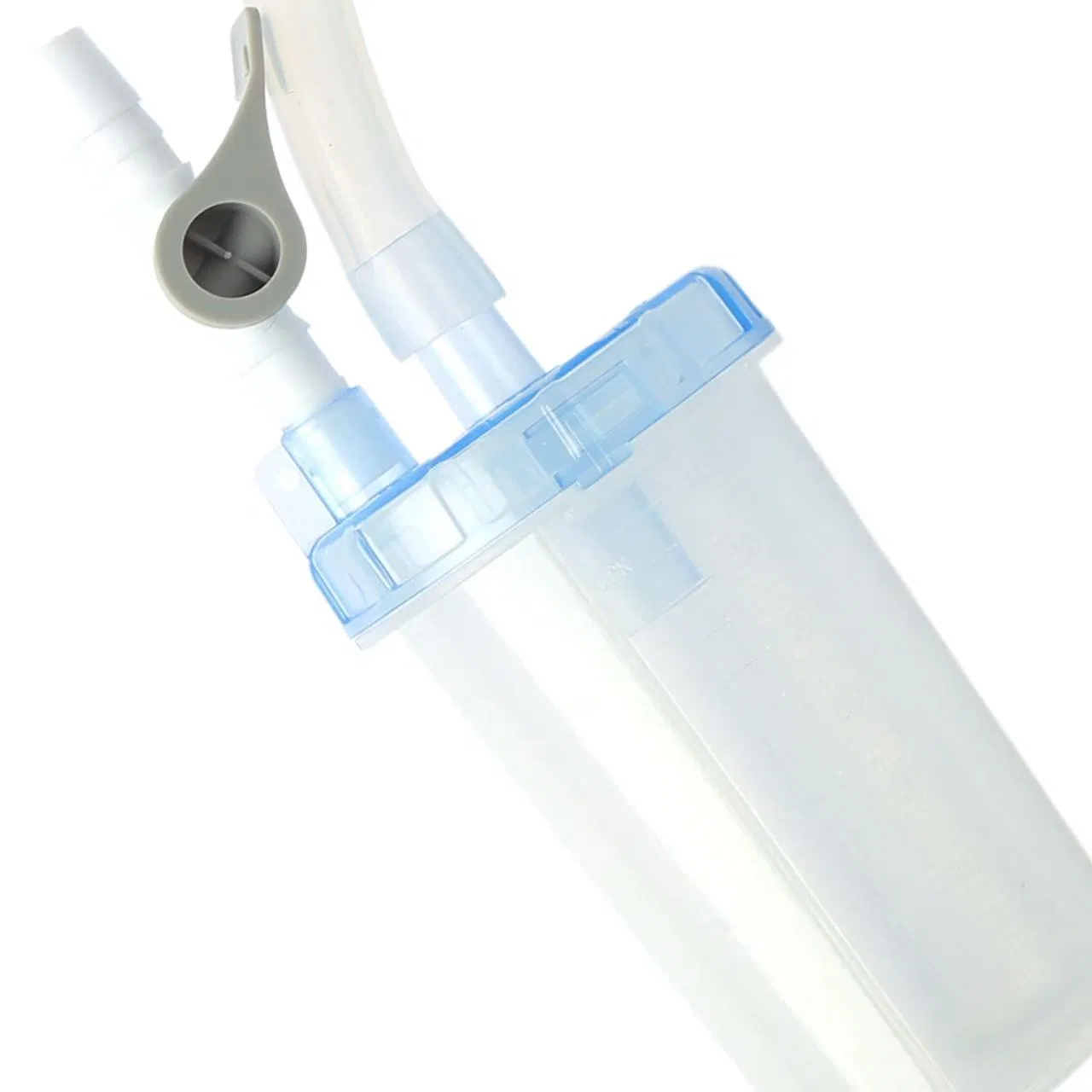
Negative Pressure Drainage Device is composed of a barrel, filter screen, cap, sealing ring, valve, and connecting tube.
Range of Application: Designed to serve as a negative pressure transmission medium and to direct and collect drainage fluids during clinical negative pressure drainage procedures.
The device effectively prevents cross-contamination. By connecting the barrel to the drainage system via the tubing, it enables effortless fluid extraction, reduces clinical procedure time, and improves workflow efficiency.
The transparency of the negative pressure drainage device allows healthcare professionals to visually monitor the characteristics of drained fluids in real-time, supporting clinical assessment.
Must be operated by trained medical professionals within healthcare facilities.
Do not use if the package is broken or tampered with.
Attention should be paid to whether the drainage is unobstructed. Rinse timely if a blockage occurs.
Post-application waste must be treated strictly according to Medical Waste Management Measures.




 Ryu Medical
Ryu Medical