













| Customization: | Available |
|---|---|
| Product Name: | Negative Pressure Drainage Device |
| Keywords: | Keywords |

| Model NO. | Negative Pressure Drainage Device |
| Sterilization | Ethylene Oxide Sterilization |
| Length | 300mm-600mm |
| Specifications | 12×300, 12×350, 12×400, 12×450, 12×500, 12×550, 12×600 |
| Capacity | 140ml |
| Certification | ISO13485 |
| Instrument Classification | Class II |
| Term of Validity | 3 Years |
| Usage | Minimally Invasive Surgical Materials |
| Packing | Plastic Package and Carton |
| Age Group | Children and Adults |
| HS Code | 9018390000 |

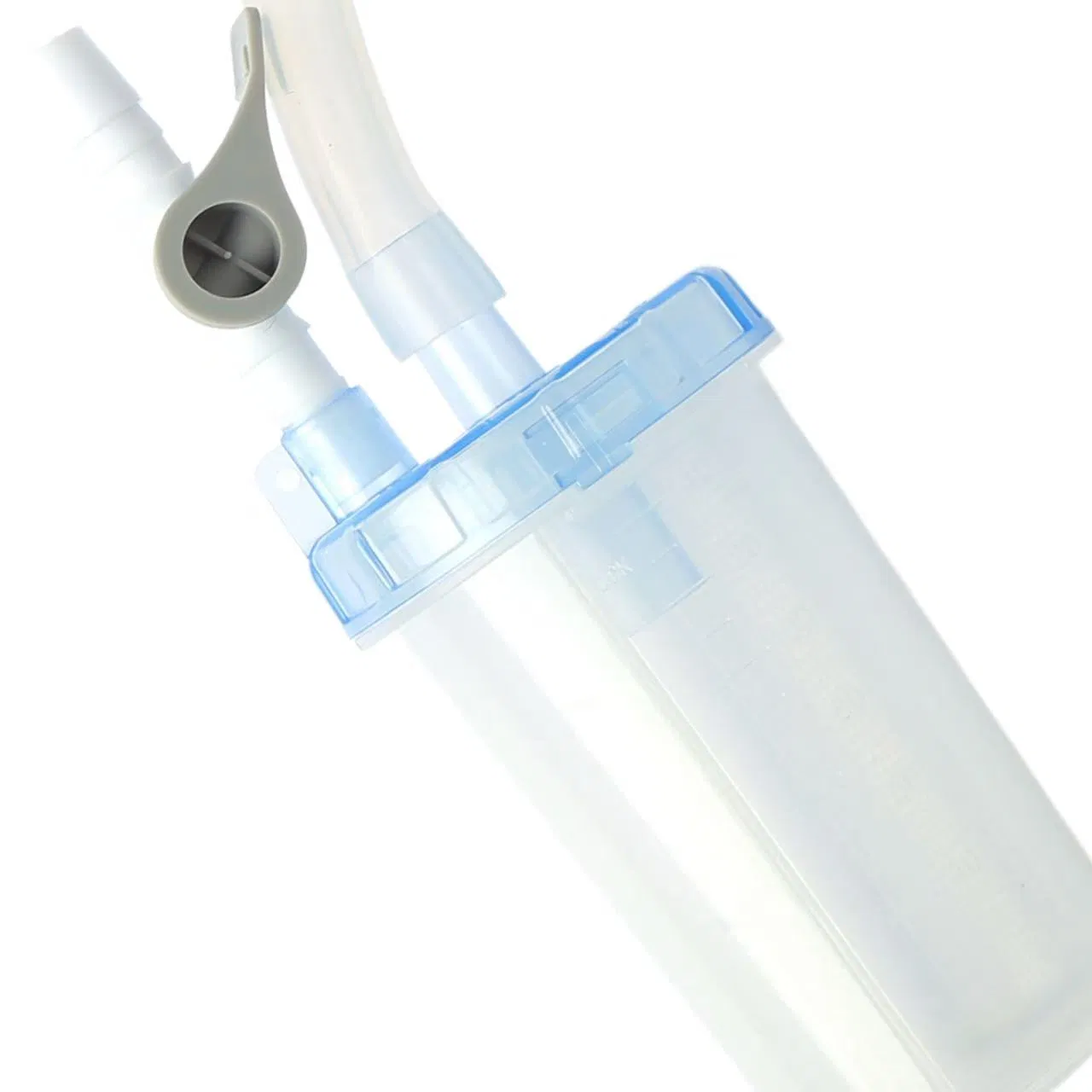
The Negative Pressure Drainage Device is a medical-grade system composed of a high-quality barrel, filter screen, secure cap, sealing ring, valve, and connecting tube. It is engineered for precision and safety in clinical environments.
This product is specifically designed to serve as a reliable negative pressure transmission medium, directing and collecting drainage fluids during clinical negative pressure drainage procedures.

Effectively prevents cross-contamination. By connecting the barrel to the drainage system via the tubing, it enables effortless fluid extraction, reduces clinical procedure time, and improves workflow efficiency.

The transparency of the device allows healthcare professionals to visually monitor the characteristics of drained fluids in real-time without opening the system, supporting rapid clinical assessment.




 Ryu Medical
Ryu Medical