














| Still deciding? Get samples of $ ! US$ 499/Piece Request Sample |
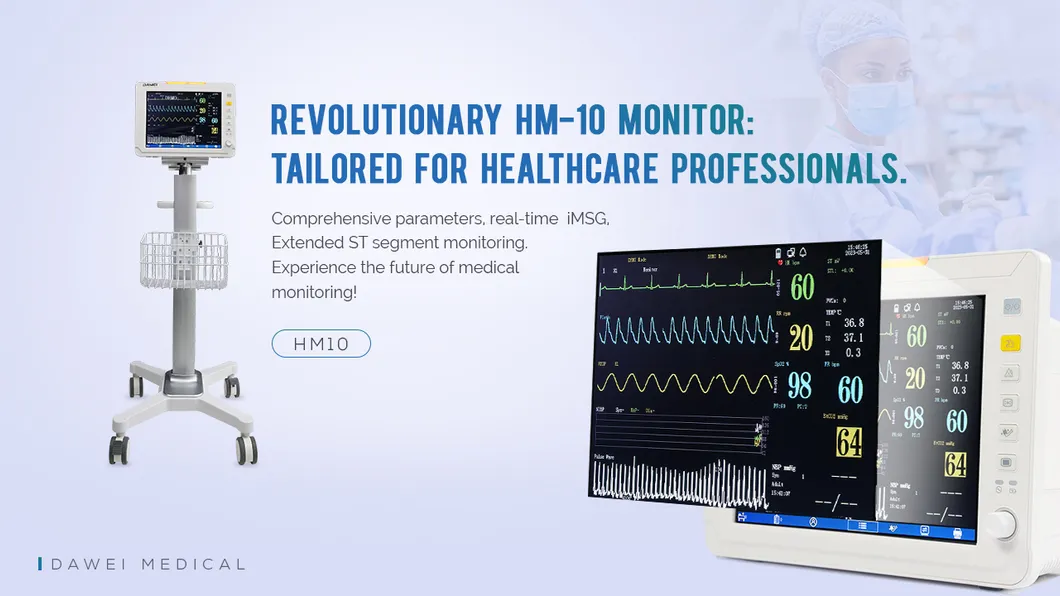
ICU Room Touch Screen Medical Multi-Parameter Portable Patient Monitor System

Standard Configuration:
ECG, Blood Oxygen, Blood Pressure, Respiratory Rate, Body Temperature, Heart Rate.
Optional Configuration:
Main stream/side stream ETCO2, IBP, etc.
Standard Accessories:
Five-lead wire, temperature probe, SpO2 probe, NIBP cuff & tube, power cord, electrode pads.



ISO 13485 certification is an international standard for the medical device industry. This emphasizes the maintenance of a quality management system to ensure that products meet rigorous industry requirements, consistently providing high-quality, safe, and reliable medical devices. Our production processes undergo rigorous monitoring to ensure product consistency and compliance with regulatory requirements.
We are a high-tech medical device manufacturer dedicated to innovative development since 2006. Our product portfolio includes ultrasound imaging systems, digital electrocardiographs, patient monitors, and digital radiography equipment. All products have received the medical device registration certificate (CFDA), European CE certification, and ISO 13485 quality system certification.



Our dedicated technical experts provide prompt assistance via phone, email, or remote assistance. We offer troubleshooting, software support, and equipment operation training. When necessary, on-site repair services are available to ensure minimal downtime for your healthcare facility.
 Ryu Medical
Ryu Medical