1 / 5










| Still deciding? Get samples of $ ! US$ 0.1/Piece Request Sample |
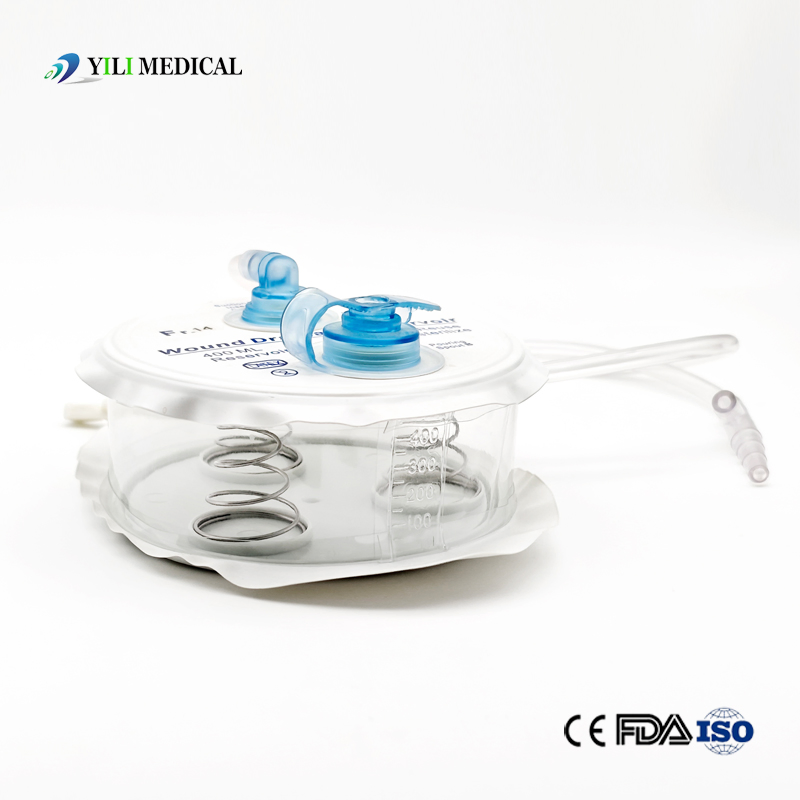
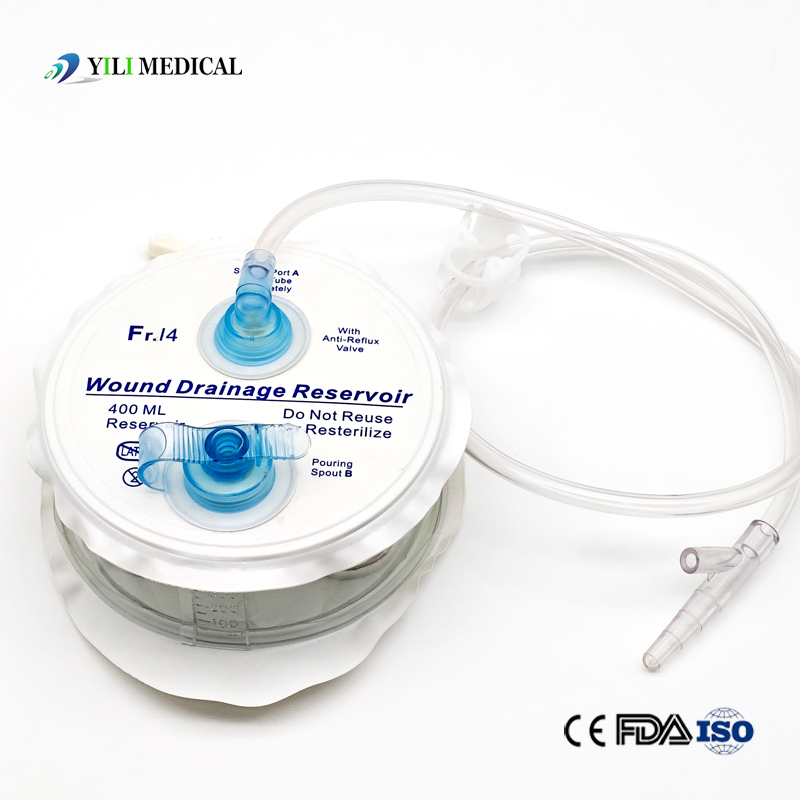
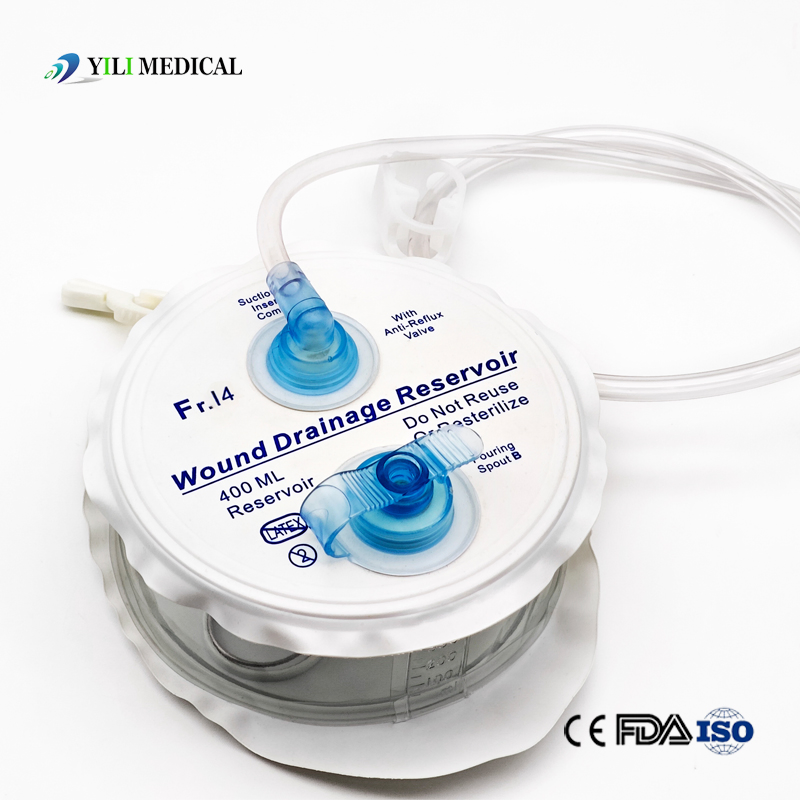
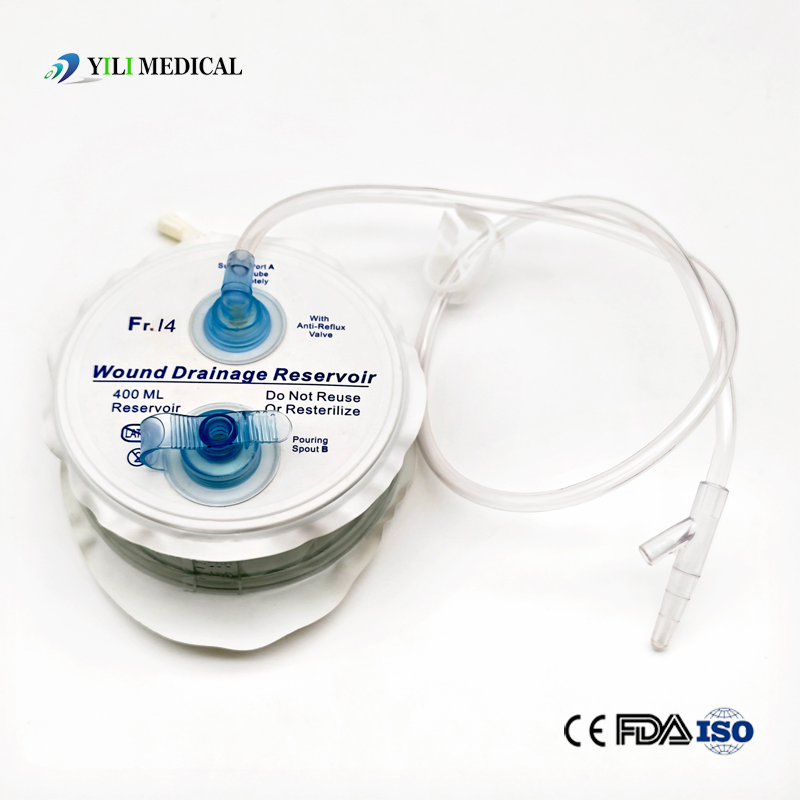
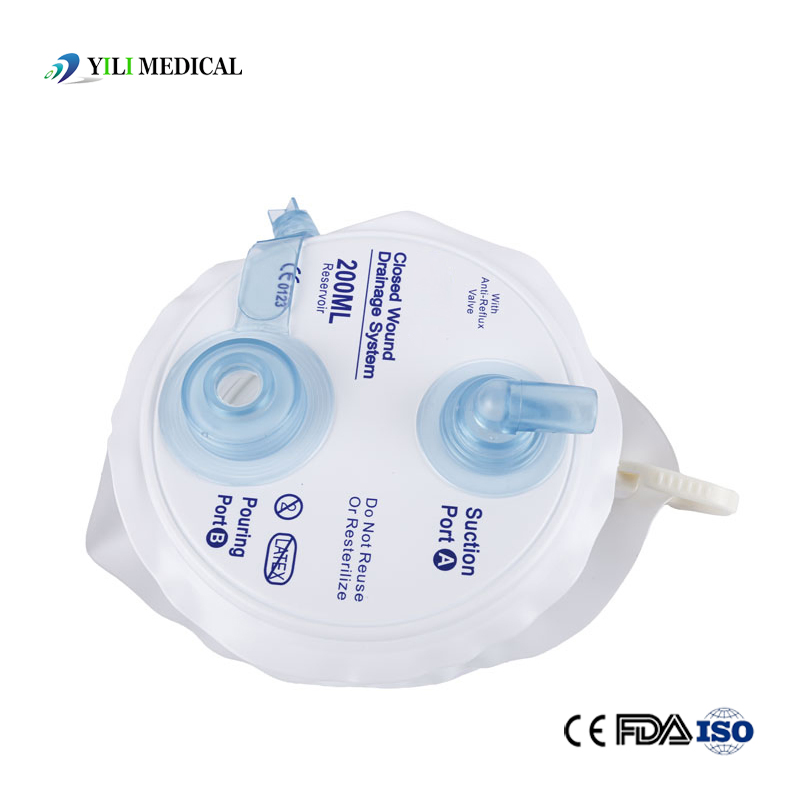
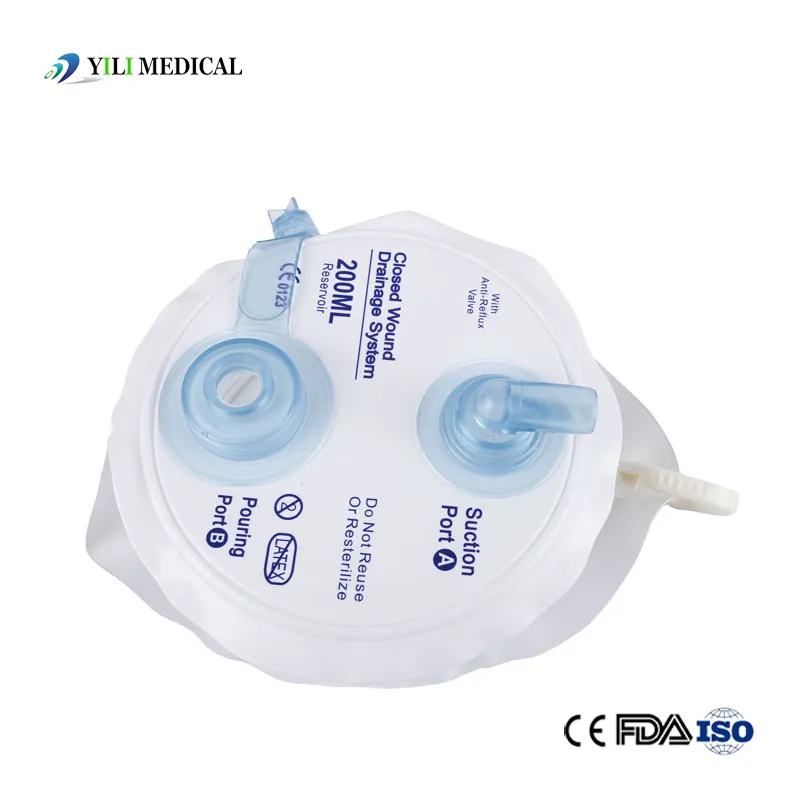
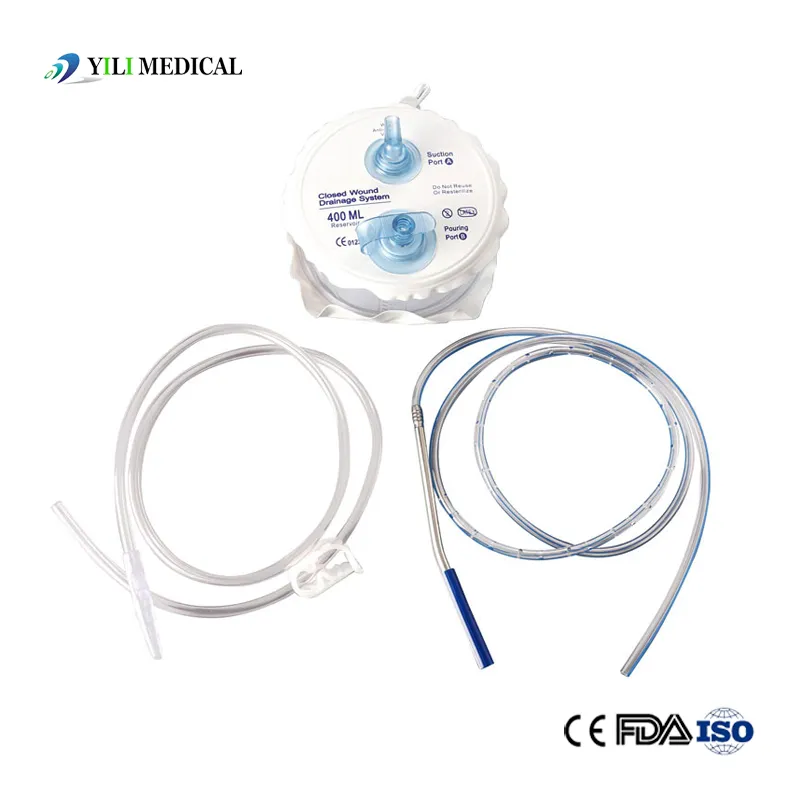
Medical Silicone Negative Pressure Closed Wound Drainage System
Operating Instructions:
| Product Name | Closed Wound Drainage System (Spring type) |
| Material | Medical Grade PVC & Silicone |
| Capacity | 100ml, 200ml, 400ml, 600ml, 800ml, 1000ml (Customizable) |
| Type | Spring type or Hollow type |
| Packing | Individual Sterile Packing |
| Sterilization | EO Gas |
| Shelf Life | 3 Years |
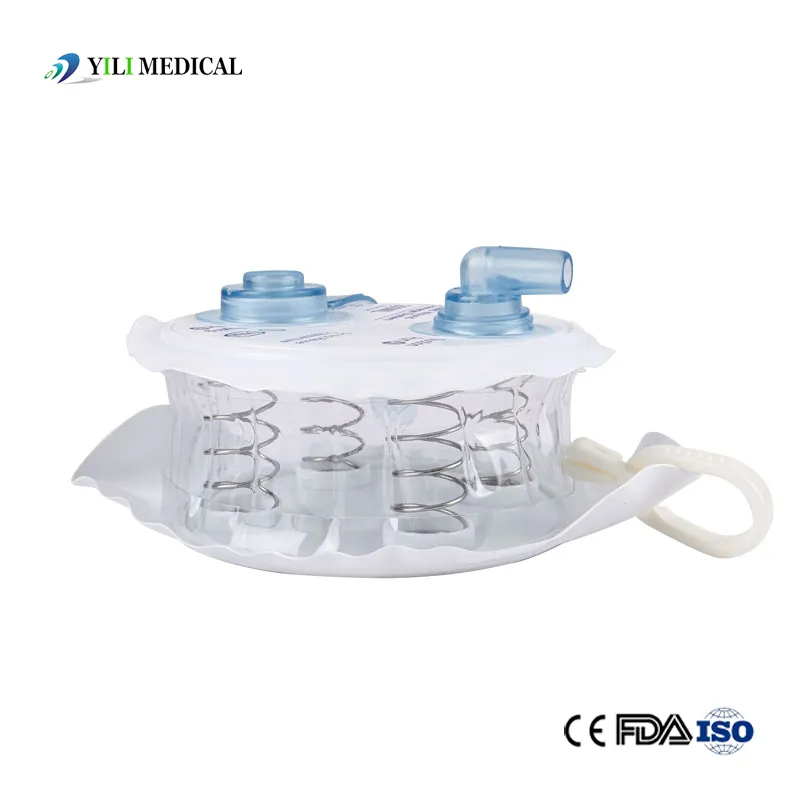
Our manufacturing processes operate under a strict ISO 13485 quality control system. All products are CE certified and produced in cleanroom environments. Each stage of production follows Standard Operating Procedures (SOP) to ensure traceability and reliability.







 Ryu Medical
Ryu Medical